Iron cathode reaction11/11/2023 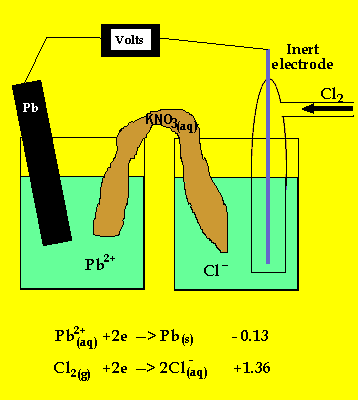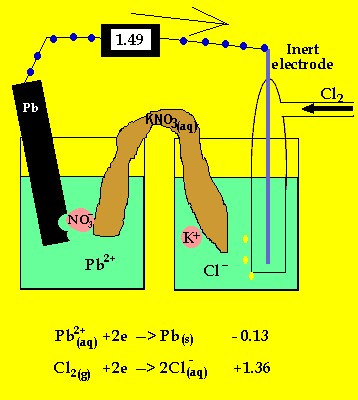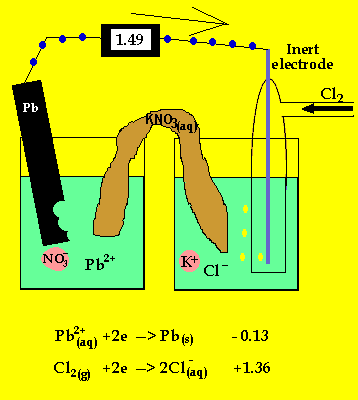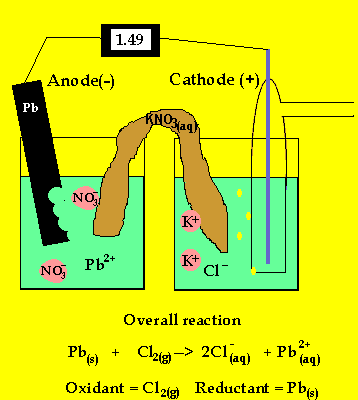 
The paint acts as a protective layer, allowing no room for oxygen to enter. This is the simplest and the efficient way which is practiced in day-to-day life, everywhere around us. Like if we apply paint, it can prevent corrosion. 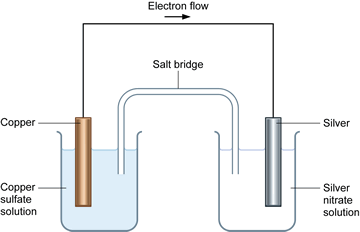
To prevent corrosion, we can apply an anti-corrosive layer of another material which can or cannot be a metal. Note: We should remember that the oxidation of many metals is a thermodynamic spontaneous process except for gold and platinum. During rusting of iron, the reaction taking place at cathode isFe2++. The electrons in the solute flows from anode to cathode through metals, while the ions flow through water droplets.įrom this study, we can observe that in the iron corrosion process, all the processes take place. Solution For During rusting of iron, the reaction taking place at cathode. The reaction taken place at the cathode is the. When iron corrodes, it forms a reddish-brown coloured metal oxide which is represented by a chemical formula - $F$before forming rust. iron cathode, with sodium chloride solution as the electrolyte. Thus, the resulting electrochemical potential develops an electric current that dissolves the less active metal electrochemically. When aqueous solutions of ionic compounds are electrolyzed, the anode and cathode half-reactions may involve the electrolysis of either water species (H 2 O, H +, OH ) or solute species (the cations and anions of the compound). The hydrogen gas is formed on the more noble or less active metal. At the anode electrons are lost from the metal, indicating an oxidation reaction, and at the cathode, electrons are gained, indicating a reduction reaction. The Electrolysis of Aqueous Sodium Chloride. We know that corrosion generally occurs when two dissimilar metals are in contact with each other in the presence of electrolyte like salt water. Applying an external potential of about 1.71.9 V to two inert electrodes immersed in an aqueous solution of an electrolyte such as H 2 SO 4 or Na 2 SO 4 drives the thermodynamically nonspontaneous decomposition of water into H 2 at the cathode and O 2 at the anode. As the time goes on, the cathode is more alkalinized and passivated by oxygen and alkali (base, OH ions) formed in the cathode reaction, but the anode is more activated by anions, e.g., chloride ions present initially in the solution, migrating through the solution with the corrosion current towards the anode, and at the same time, by. At cathode, reduction takes place, so reaction will be. Galvanic corrosion is an electrochemical process of erosion of metals. The ions are connected by a salt bridge, separated by a porous membrane. It is a process through which metals deteriorate when exposed to oxygen.Ī galvanic cell consists of two different metals immersed in electrolytes of the same metals in aqueous solution. Here, they form a solid called hydrated iron oxide, 2Fe 2 sub)3 sub)2 The water layer on the surface of the iron acts as a barrier, or salt bridge, that is needed for the electrochemical cell or. It is an electro-chemical cell that generates electrical energy from spontaneous redox reactions taking place inside the cell. =1.23\ V\).Hint: We have studies about galvanic cells. The position of tin and iron in the electro-chemical series indicates that tin (-0.14V) will act as the cathode and iron (-0.44V) as the anode in the local.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
